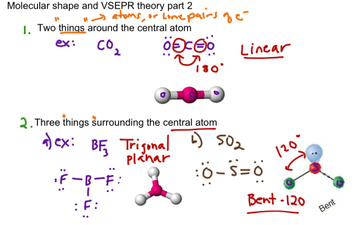
10.3 Bond Dissociation Energy Bond Length H 2 F 2 436.4 kJ/mole 150.6 kJ/mole 74 pm 142 pm Overlap Of 2 1s 2 2p How does Lewis theory explain the bonds in H 2 and F 2 ? Sharing of two electrons between the two atoms. Valence bond theory – bonds are formed by sharing of e - from overlapping atomic orbitals.10.2 Chemistry In Action: Microwave Ovens.10.2 dipole moment polar molecule no dipole moment nonpolar molecule dipole moment polar molecule no dipole moment nonpolar molecule Which of the following molecules have a dipole moment? H 2 O, CO 2, SO 2, and CH 4 O H H S O O C O O C H H H H.Dipole Moments and Polar Molecules 10.2 electron rich region electron poor region H F .VSEPR 10.1 AB 4 E 2 4 2 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 6 6 0 octahedral octahedral AB 5 E 5 1 octahedral square pyramidal octahedral square planar Xe F F F F.VSEPR 10.1 AB 5 E 5 1 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 6 6 0 octahedral octahedral octahedral square pyramidal Br F F F F F.VSEPR 10.1 AB 5 5 0 trigonal bipyramidal trigonal bipyramidal AB 2 E 3 2 3 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 4 E 4 1 trigonal bipyramidal See-Saw AB 3 E 2 3 2 trigonal bipyramidal T-shaped trigonal bipyramidal linear I I I.VSEPR 10.1 AB 5 5 0 trigonal bipyramidal trigonal bipyramidal AB 3 E 2 3 2 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 4 E 4 1 trigonal bipyramidal See-Saw trigonal bipyramidal T-shaped Cl F F F.VSEPR 10.1 AB 5 5 0 trigonal bipyramidal trigonal bipyramidal AB 4 E 4 1 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry trigonal bipyramidal See-Saw (distorted tetrahedron).VSEPR AB 4 4 0 tetrahedral tetrahedral 10.1 AB 2 E 2 2 2 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 3 E 3 1 tetrahedral trigonal pyramidal tetrahedral bent H O H.VSEPR AB 3 E 3 1 AB 4 4 0 tetrahedral tetrahedral 10.1 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry tetrahedral trigonal pyramidal. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
